Abstract
Background and Aim: Wound healing after proctological surgery in the anal and perianal region may be compromised due to the anatomical and functional characteristics of this area; microbiological contamination and defecation-related mechanical stress are key factors that delay local reparative processes. This review aims to systematize the systemic, local, and procedure-specific risk factors responsible for delayed epithelialization of postoperative anal and perianal wounds, to interpret the underlying pathophysiological mechanisms from a practical perspective, and to present a stepwise, evidence-based management approach. Methods: A narrative review of recent literature from PubMed, Scopus, and major international guidelines (ASCRS, ESCP, EWMA, WHO) was performed. Data regarding pathogenesis, risk factors, diagnostic criteria, and contemporary treatment strategies for non-healing perianal wounds were analyzed and synthesized. Results: Delayed epithelialization is a multifactorial process resulting from the synergistic effects of local factors (persistent bacterial burden, tissue hypoxia, mechanical stress, and moisture), systemic conditions (diabetes, malnutrition, anemia, immunosuppression, smoking), and procedure-related determinants (extent of tissue trauma, wound configuration, residual sphincter spasm). Perianal wounds frequently evolve toward a chronic wound phenotype, rendering standard local care insufficient. A substantial proportion of cases are linked to inadequate initial pathogenetic and differential diagnostics, particularly failure to exclude inflammatory bowel disease. Contemporary evidence supports a multimodal strategy combining optimization of the local wound environment, correction of systemic risk factors, and selective use of adjunctive technologies such as NPWT. Conclusions: Delayed epithelialization of anal and perianal wounds represents a dynamic and modifiable biological process rather than a static postoperative complication. Early identification of risk factors, pathogenetically oriented diagnostics, and stepwise, individualized management can significantly reduce postoperative morbidity, shorten healing time, and improve patient quality of life.
Full article
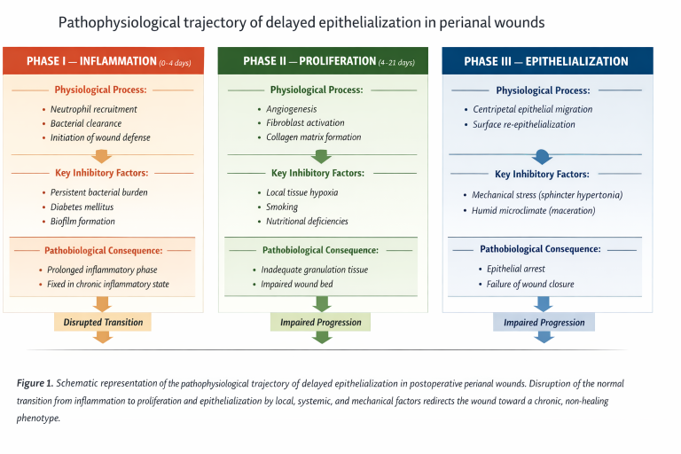
Introduction: Wound healing after proctological surgery in the anal and perianal region is associated with specific challenges arising from the anatomical and functional characteristics of this area. Constant microbiological contamination, repetitive mechanical microtrauma during defecation, the humid perianal environment, and the dynamic activity of the sphincter complex can significantly alter the normal course of reparative processes [1–5]. Consequently, a proportion of postoperative wounds exhibit delayed epithelialization, prolonged exudation and pain, and in some cases the development of persistent sinus or fistulization and chronic wound defects [2,3,5,6]. Complex perineal wounds not only increase patient discomfort but are also associated with additional interventions, long-term dressing requirements, and increased utilization of healthcare resources [1,2,4]. Contemporary concepts emphasize that delayed epithelialization is not a “single-cause” phenomenon but rather a multifactorial process. Local microcirculatory disturbances, persistent bacterial burden and subclinical infection, tissue tension, and impaired progression of the granulation phase may act simultaneously with patient-related systemic factors such as metabolic disorders, poor nutritional status, and impaired immune response [3–5,7]. In this context, recent years have witnessed growing attention to the development of structured diagnostic and therapeutic algorithms for chronic postoperative wounds of the anal canal and perineum, the establishment of practical criteria for defining “non-healing wounds,” and the selection of targeted stages of intervention [3,4,9–11]. The literature identifies two principal pillars of management strategies: (1) systemic optimization and prevention of complications, including correction of risk factors, timely recognition of infection, and management of adverse events [3,4,8]; and (2) stage-adapted individualization of local therapy, encompassing the selection of dressings that maintain moisture balance, topical agents that stimulate granulation and reduce bacterial load, and, in selected cases, negative pressure wound therapy (NPWT) and other adjunctive modalities [3,4,9–11]. For example, randomized clinical data indicate that the addition of topical agents to standard care in infected wounds after anorectal surgery improves healing outcomes, thereby substantiating the concept of “adjuvant local therapy” in postoperative perianal wounds [11,12]. Conversely, in challenging clinical scenarios such as extensive perineal defects and persistent perineal sinus, management should not be limited to simple “dressing changes.” Contemporary reviews discuss the potential role of NPWT, hyperbaric oxygen therapy, flap reconstruction, and regenerative approaches in selected patient groups [3,4,9,13,14]. Furthermore, recent studies have separately evaluated the effects of active wound dressings on pain and exudation, as well as their potential to enhance healing efficiency [11,12,15]. In this narrative review, systemic, local, and procedure-specific risk factors contributing to delayed epithelialization of postoperative anal and perianal wounds are systematized; the pathophysiological mechanisms underlying impaired healing are interpreted from a practical perspective; and a stage-based management approach grounded in current evidence is presented.
Pathogenesis and Risk Factors of Delayed Epithelialization in Postoperative Anal and Perianal Wounds: Delayed epithelialization of postoperative anal and perianal wounds is rarely attributable to a single mechanism; rather, it develops as a result of the complex interplay between local, systemic, and procedure-specific factors (Fig 1) [1–4,7,16]. Although normal wound healing requires the sequential and balanced progression of the inflammatory, proliferative, and epithelialization phases, this orderly process is frequently disrupted in the perianal region [1,3,4]. Consequently, the determinants of delayed healing can be conventionally classified into three principal categories: local, systemic, and procedure-specific factors [2,4,7].
Local Factors: The anal and perianal region is regarded as one of the most anatomically and biologically complex areas in terms of wound healing [1–5]. In this zone, delayed epithelialization is determined not only by mechanical influences but also by the specific pathophysiological characteristics of the local tissue environment [3,5,17]. In the perianal area, the reparative potential of a wound is constrained by several parallel mechanisms, which promote prolongation of the inflammatory phase and facilitate the development of a chronic wound phenotype [3–5,17]. One of the principal local determinants is the persistent microbiological burden. The anal canal and surrounding skin are in continuous contact with colonic flora, making bacterial colonization of an open wound surface virtually unavoidable [1–3,5,17]. This colonization not only increases the risk of infectious complications but also sustains prolonged activation of the inflammatory cascade. Continuous neutrophil recruitment and high concentrations of proteolytic enzymes inhibit epithelial cell migration and fibroblast proliferation [3,5,17]. With prolonged colonization, the likelihood of biofilm formation increases, thereby reducing the effectiveness of both local antiseptics and systemic antibiotics and leading to “fixation” of the wound in the inflammatory phase [11,17]. Local microcirculatory impairment and tissue hypoxia constitute another key mechanism. Chronic anal fissure, prolonged sphincter spasm, and repeated surgical interventions induce structural alterations in the submucosal vascular network [1,3,5,18]. Postoperative edema, venous congestion, and increased interstitial pressure further compromise capillary perfusion [3–5]. In a hypoxic microenvironment, angiogenesis is attenuated, fibroblast metabolic activity declines, and the formation of the collagen matrix is delayed. As a result, the wound encounters difficulty transitioning into the proliferative phase and remains trapped in the inflammatory stage for a prolonged period [3–5,11]. Another fundamental mechanism disrupting epithelialization in the perianal region is mechanical tension and recurrent microtrauma. During defecation, traction forces generated at the wound margins mechanically interfere with the centripetal migration of epithelial cells from the periphery toward the center of the defect [1–4]. Newly formed, fragile granulation tissue is easily damaged, microscopic fissures reopen, and the reparative process is repeatedly “reset” to its initial stage [3,5]. This effect is particularly pronounced in patients with sphincter hypertonia; even after lateral internal sphincterotomy, the persistence of residual spasm may sustain mechanical stress and continue to impede healing [5,18]. In addition, the humid microclimate of the perianal region compromises the stability of the epithelial barrier. Persistent moisture and secretion lead to maceration of the skin, reduction of the protective function of the stratum corneum, and creation of a favorable environment for secondary microbial invasion. Under these conditions, the wound edges become fragile, cellular migration assumes an unstable pattern, and the continuity of epithelialization cannot be maintained [3,5,11,17]. Thus, the synergistic effects of microbiological contamination, perfusion deficits, mechanical stress, and a persistently moist environment fundamentally alter the biological behavior of perianal wounds, distancing them from the classical “acute wound” model and shifting them toward the pathophysiology of chronic wounds. This underscores that delayed epithelialization is not merely a consequence of “inadequate wound care” but reflects the unique local biology of the perianal region, thereby necessitating individualized therapeutic strategies tailored to this specific anatomical and physiological context.
Systemic Factors: Wound healing is not merely a local tissue response but a complex biological process reflecting the overall reparative capacity of the organism. Following surgical interventions in the anal and perianal region, epithelialization of the wound is closely associated with the patient’s general somatic condition, and disturbances at the systemic level may substantially diminish the effectiveness of local therapeutic measures [8,16]. Diabetes mellitus and metabolic dysfunction represent leading determinants of impaired wound healing. Diabetes-associated microangiopathy compromises tissue perfusion, resulting in reduced delivery of oxygen and nutrients at the capillary level. Concurrently, diabetic neuropathy diminishes sensory perception in the perianal region, allowing microtrauma to go unnoticed and mechanical stress to persist. Under hyperglycemic conditions, the functional activity of neutrophils and macrophages is impaired, bacterial clearance is reduced, and the inflammatory phase is prolonged. The convergence of these mechanisms delays progression to the proliferative and epithelialization phases of wound repair [8]. Nutritional deficiency and hypoproteinemia undermine the biochemical foundations of the reparative process. Deficiencies of protein, zinc, iron, and vitamin C restrict fibroblast proliferation, collagen synthesis, and extracellular matrix remodeling. Reduced serum albumin levels disrupt interstitial fluid balance, promote tissue edema, and diminish the local bioavailability of therapeutic agents. Under these conditions, the proliferative potential of the wound is markedly impaired [8,10]. Anemia and systemic hypoxia restrict tissue oxygenation, thereby inhibiting the fundamental mechanisms of angiogenesis and cellular proliferation. Under conditions of oxygen deprivation, fibroblast metabolism is attenuated, collagen matrix formation becomes inadequate, and epithelial cell migration is delayed. When these effects are superimposed on the pre-existing local hypoxia of the perianal region, their clinical impact becomes particularly pronounced [16]. Immunosuppression and chronic inflammatory conditions—including autoimmune disorders and prolonged corticosteroid therapy—disrupt regenerative mechanisms at the cellular level. The transition of macrophages into the reparative phase is delayed, cytokine balance remains skewed toward a pro-inflammatory profile, and the wound becomes “fixed” in the inflammatory stage. This mechanism is especially evident in perianal defects associated with inflammatory bowel disease, where impaired healing is frequently observed in clinical practice [16,19]. Cigarette smoking constitutes an independent risk factor for delayed healing in perianal wounds. Nicotine induces peripheral vasoconstriction, while carbon monoxide reduces the oxygen-carrying capacity of hemoglobin. As a consequence, tissue hypoxia is exacerbated, fibroblast functional activity declines, and the rate of epithelialization is diminished [10,19].
These systemic factors act synergistically with local mechanisms, altering the biological behavior of the wound and reclassifying it as a “high-risk wound.” In such patients, standard local wound care strategies are frequently insufficient, and effective management must also incorporate optimization at the systemic level.
Procedure-Specific Factors: The course of postoperative epithelialization is determined not only by the patient’s biological characteristics but also by the nature and technical features of the surgical intervention itself. The initial wound environment created during surgery defines the starting trajectory of the reparative process [1,2,4]. Procedures associated with extensive tissue defects—such as conventional hemorrhoidectomy, wide fistulotomy, and open drainage of perianal abscesses—produce wounds that “secondary intention healing”. These wounds are inherently characterized by a prolonged inflammatory phase and, under the influence of the perianal environment, demonstrate a greater propensity for chronicity. The presence of a large open tissue bed, continuous microbiological contamination, mechanical stress and microtrauma during defecation, sphincter dynamics, and a persistently humid microclimate constitute the principal mechanisms underlying delayed epithelialization in such settings [1,2,4]. Excessive tissue trauma and suboptimal hemostasis contribute to deepening of local edema, elevation of interstitial pressure, and impairment of capillary perfusion. Under these conditions, a hypoxic microenvironment develops and transition to the proliferative phase is delayed. Simultaneously, hematomas and residual necrotic tissue serve as favorable substrates for bacterial colonization [2,4]. Interventions involving the sphincter apparatus exert a specific influence on epithelialization. Although the primary objective of lateral internal sphincterotomy is to relieve anal hypertonia, residual spasm or the predominance of fibrotic sphincter tissue may persist in certain patients, perpetuating mechanical tension. In this context, traction at the wound margins is maintained, epithelial cell migration is mechanically constrained, and the healing process is delayed. In addition, posterior internal sphincterotomy may be associated with the development of keyhole deformity, which can lead to persistent soiling, moisture imbalance, and impaired wound edge integrity. In such cases, altered local biomechanics and chronic contamination may further delay epithelialization and contribute to a non-healing wound phenotype [18]. The localization of the operation and wound configuration also play a critical role. Defects situated along the posterior midline are located in anatomically less well-perfused zones and therefore exhibit a lower propensity for epithelialization. Deep, narrow wounds with inadequate ventilation favor accumulation of exudate and formation of a persistently moist microenvironment, conditions that promote biofilm development and prolongation of the inflammatory phase, ultimately fostering a chronic wound phenotype [1,4,17]. Thus, the extent of surgical intervention, degree of tissue trauma, wound configuration, and functional status of the sphincter apparatus represent the principal procedure-specific determinants shaping the trajectory of postoperative epithelialization. Accurate assessment of these factors enables early prediction of delayed healing risk and facilitates the development of individualized postoperative management strategies.
Clinical Consequences of Delayed Epithelialization and Diagnostic Approach: Delayed epithelialization of postoperative anal and perianal wounds represents not merely a local morphological issue but a clinical syndrome that directly affects the patient’s quality of daily life, functional capacity, and psychological well-being. Persistent pain, burning and discomfort, continuous serous or purulent discharge, and fear and tension during defecation impair social adaptation and may reduce adherence to treatment. In clinical practice, such conditions frequently lead to repeated consultations, additional interventions, and, at times, unjustified antibiotic use.
Over time, a non-healing wound may give rise to the following complications:
• formation of a chronic inflammatory focus,
• persistence of a perianal sinus or secondary fistulization,
• fibrosis and excessive scar tissue formation,
• in rare cases, keloid-type scar changes,
• chronification of pain syndrome and defecatory dysfunction.
From this perspective, early recognition of delayed epithelialization and accurate differential diagnosis are of critical clinical importance [1,2,4].
Clinical Criteria: In practical terms, a postoperative wound may be considered “delayed-healing” under the following conditions:
• failure to achieve complete epithelialization more than six weeks after surgery;
• poor development or a “pale” appearance of granulation tissue on the wound surface;
• persistent serous or mild purulent discharge;
• ongoing pain and burning during defecation;
• fibrosis and reduced elasticity at the wound margins.
Although these features do not, in isolation, prove the presence of infection, they indicate deviation from the normal reparative stages and necessitate further evaluation [3,4].
Differential Approach: Delayed epithelialization must be distinguished from the following clinical conditions:
• residual or newly formed anal fissure,
• subclinical abscess or microinfection,
• persistent sinus tract,
• early fistula formation,
• secondary wound changes associated with allergic or contact dermatitis.
This differentiation is fundamental for selecting the appropriate therapeutic strategy, as management differs substantially between simple delayed healing and structural pathology [2,4].
Diagnostic Algorithm:The diagnostic approach should be stepwise and goal-oriented:
1. Clinical inspection and palpation osize, depth, and condition of wound margins; ocharacteristics of granulation tissue;
otype and amount of exudate; osigns of maceration and dermatitis in the perianal skin [18,19].
2. Assessment of sphincter tone persistent hypertonia may represent the principal source of mechanical stress; residual spasm should be considered even after LIS [18]. in selected cases, functional assessment may be complemented by anorectal manometry[19] .
3. Instrumental evaluation (in selected cases) endoanal ultrasound in suspected microabscess, sinus, or early fistula; magnetic resonance imaging in extensive and complex perineal defects [19]. anorectal manometry in selected cases to assess sphincter function, particularly in patients with persistent hypertonia or suspected functional abnormalities contributing to delayed healing [18].
4. Laboratory assessment
o hemoglobin, glucose, C-reactive protein;
o albumin and total protein levels;
o microbiological analysis of wound discharge when infection is suspected [3,8–11,16].
This algorithm enables differentiation between simple reparative delay and pathology associated with structural disease, thereby facilitating rational planning of subsequent therapeutic stages.
Common Diagnostic Errors in Clinical Practice and Their Association with Delayed Epithelialization: One of the principal causes of delayed healing of postoperative anal and perianal wounds is not the surgical technique itself, but rather inadequate diagnostic assessment at the initial stage. In routine clinical practice, anal fissures and perianal fistulas are often regarded as isolated local pathologies, while the pathogenic nature of the process is relegated to secondary importance. This approach prevents timely identification of the underlying etiological factor—most notably inflammatory bowel disease—in a subset of patients [18,19]. One of the most frequent errors is the failure to routinely consider colonoscopy in patients presenting with anal fissures or fistulas. However, in the following clinical scenarios, this examination is not merely desirable but pathogenetically essential:
• atypically located anal fissures in young patients,
• multiple or complex perianal fistulas,
• recurrent abscess formation,
• disproportionate pain and prolonged discharge,
•presence of systemic features (weight loss, chronic diarrhea, subfebrile temperature).
This constellation of findings may be characteristic of the perianal manifestation of Crohn’s disease. When surgical intervention in such patients is performed solely as a mechanical solution and the underlying disease is not addressed, the biological behavior of the wound changes: inflammation persists, granulation remains inadequate, and epithelialization is delayed. Consequently, even a technically correct operation may culminate in a non-healing wound [19]. Pathogenetic and differential diagnostic approaches constitute a cornerstone of modern management of anal and perianal pathology. Anal fissures and fistulas should not be regarded merely as morphological defects but, in certain cases, as local manifestations of systemic disease. Accordingly, in patients at risk:
• performance of colonoscopy,
• macroscopic and microscopic evaluation of the intestinal mucosa,
• exclusion of Crohn’s disease, ulcerative colitis, and other inflammatory conditions play a critical role in preventing postoperative complications, particularly delayed epithelialization [18,19]. Thus, a proportion of non-healing perianal wounds arise not from deficiencies in surgical technique, but from failure to appropriately assess the pathogenic nature of the disease at an early stage. Contemporary proctological practice requires that anal and perianal pathologies be approached not solely as local anatomical problems, but as potential indicators of systemic disease. Implementation of this principle may substantially reduce the incidence of delayed wound healing, as well as recurrence and chronicity.
Contemporary Treatment Strategies and Stepwise Management: Cases of delayed epithelialization following anal and perianal surgery cannot be managed within the framework of a passive “wait-and-heal” approach. In such situations, therapeutic strategy must be aligned with the wound’s pathobiology and structured in a stepwise, individualized manner. The primary objectives are to interrupt the inflammatory phase, stimulate transition to the reparative stage, and prevent transformation of the wound into a “chronic wound phenotype.” Contemporary management is based on three fundamental pillars:
1. optimization of the local wound environment,
2. correction of systemic factors,
3. application of invasive or technological interventions in selected cases [3,4].
Local Treatment and Optimization of the Wound Environment: In perianal wounds, the principal therapeutic goal is to reduce inflammatory burden, normalize moisture balance, and create a microenvironment favorable for epithelial cell migration [3,9–11].
Principle of Controlled Moisture Balance: Modern wound care has abandoned the concept of “complete drying.” Maintenance of optimal moisture accelerates epithelial cell migration, facilitates separation of necrotic tissue, and reduces pain. Although preserving this balance in the perianal region is challenging, it is achievable through appropriate local measures [11,15].
Daily Hygiene and Reduction of Mechanical Stress: Warm sitz baths, gentle cleansing, and elimination of irritative factors (hard stools, excessive friction) attenuate the local inflammatory response [10,16]. Softening of defecation reduces repetitive microtrauma at the wound margins and ensures continuity of the healing process [3,18].
Topical Agents:Stage-adapted local therapies may direct reparative processes:
• short courses of antimicrobial gels or ointments in the presence of increased bacterial load [12,22],
• vasodilatory topical preparations to alleviate pain and tension associated with sphincter hypertonia [18],
• regenerative agents that stimulate granulation and support epithelialization [3,11].
In patients with a tendency toward keloid or hypertrophic scarring, application of silicone-based gels to the perianal skin after completion of epithelialization may help prevent pathological scar thickening [10,11].
Correction of Systemic Factors: When a non-healing wound is regarded solely as a local problem, treatment is often ineffective. Therefore, in every patient, reparative capacity at the systemic level should be assessed and optimized whenever possible:
• control of hyperglycemia [8],
• restoration of albumin and total protein levels [8],
• correction of anemia,
• consideration of immunosuppressive conditions,
• smoking cessation.
These interventions are as important as measures applied directly to the wound, and their absence may markedly reduce the effectiveness of local therapy [3,4].
Technological and Invasive Approaches in Selected Cases: In situations where epithelialization does not progress despite standard local and systemic measures, particularly in the presence of exudative and extensive defects, additional modalities should be considered.
Negative-Pressure Wound Therapy (NPWT): Especially in deep, exudative, or extensive perineal wounds, low-pressure systems reduce edema, improve microcirculation, and accelerate granulation tissue formation. NPWT may alter the “biological behavior” of the wound, facilitating transition into the proliferative phase [9,13,14,20,21].
Minimal Surgical Revision: In the presence of residual necrotic tissue, persistent sinus tracts, or microabscesses, local debridement and drainage may be necessary to restore reparative processes [3,4].
Exclusion and Elimination of Structural Pathology: If delayed healing is associated with recurrent fissure, residual sphincter spasm, early fistula formation, or underlying systemic disease, limitation to wound care alone is inappropriate. In such cases, elimination of the primary pathogenetic factor (e.g., additional sphincter relaxation, specific treatment of inflammatory bowel disease) is decisive [18,19]. Thus, contemporary management conceptualizes delayed epithelialization not as a static condition but as a dynamic biological process. Optimization of the local environment, correction of systemic factors, and, in selected cases, implementation of technological interventions enable modification of the wound’s reparative trajectory and prevention of chronicity.
Discussion: The principal findings of this review indicate that delayed epithelialization of postoperative anal and perianal wounds is not merely a “wound problem,” but rather a complex pathobiological process determined by the specific characteristics of the local tissue environment, the patient’s systemic condition, and the adequacy of the initial diagnostic approach. Data presented in the literature consistently confirm the multifactorial nature of this phenomenon and emphasize that, in contrast to other surgical sites, the perianal region carries a distinct risk profile [1–4,7]. Many authors identify persistent microbiological contamination, continuous mechanical stress, and local perfusion disturbances as the primary determinants of wound chronicity in the perianal region [1–3,17]. These mechanisms prolong the inflammatory phase within the classical wound-healing model and delay transition to the proliferative stage. However, analysis of the literature demonstrates that these local factors frequently act synergistically with systemic disturbances—such as metabolic dysfunction, hypoproteinemia, anemia, and impaired immune response—thereby altering the biological behavior of the wound and rendering standard local care strategies insufficient [3,8,17].
In this context, a particularly important observation is that a substantial proportion of non-healing wounds arise not from technical surgical errors, but from incomplete pathogenetic and differential diagnostic evaluation at the initial stage. Considering anal fissures and perianal fistulas solely as local mechanical problems, especially in patients at risk, and failing to perform colonoscopy or exclude inflammatory bowel disease in a timely manner may subsequently result in delayed epithelialization, recurrence, and chronicity. The literature repeatedly underscores that when perianal Crohn’s disease is managed as “ordinary anal pathology,” surgical interventions are biologically predisposed to failure. This highlights that management of perianal wounds cannot be confined to a purely local anatomical approach [9,10]. Contemporary sources advocate for stepwise, pathogenetically grounded therapeutic strategies. Maintenance of an optimal wound microenvironment—specifically, controlled moisture of the wound surface—together with reduction of inflammatory burden and stimulation of epithelial cell migration, is regarded as a core principle [11,15]. At the same time, sustained efficacy of local interventions is unlikely without timely identification and correction of systemic risk factors [3,4,8]. In selected cases, implementation of technological approaches such as negative-pressure wound therapy and minimal surgical revision may alter the biological trajectory of the wound and redirect it toward the proliferative phase [9,13,14,20,21]. Synthesis of these approaches within the literature demonstrates that delayed epithelialization is not a static condition but a modifiable biological process. In clinical practice, it should be viewed not merely as “prolonged healing,” but as the establishment of a pathological trajectory. Integrated application of local care, systemic optimization, and an appropriate diagnostic framework may exert a tangible impact on reducing postoperative complications [1–4,13–18,21,22]. Collectively, these findings indicate that the key to preventing delayed wound healing after proctological surgery lies not solely in technical surgical excellence, but in the formation of pathogenetic clinical thinking. Approaching anal and perianal pathology as a potential local manifestation of systemic disease, integrating modern principles of wound biology into daily practice, and targeting risk factors at an early stage may substantially reduce the incidence of delayed epithelialization.
Conclusion: Delayed epithelialization of postoperative anal and perianal wounds should not be regarded merely as a local surgical problem, but rather as a complex and modifiable biological process determined by the characteristics of the local tissue environment, the patient’s systemic condition, and the adequacy of the initial diagnostic approach. Analysis of the literature demonstrates that a substantial proportion of non-healing perianal wounds arise not from technical deficiencies, but from failure to correctly assess the pathogenetic nature of the disease at an early stage. Considering anal fissures and fistulas solely as mechanical local problems, omitting colonoscopy in patients at risk, and failing to exclude inflammatory bowel disease may ultimately result in delayed healing, recurrence, and chronicity. Contemporary concepts view delayed epithelialization not as a static condition, but as a dynamic and potentially reversible biological process. Effective management should be based on three core principles:
1. optimization of the local wound environment and maintenance of controlled moisture at the wound surface;
2. timely identification and correction of systemic risk factors;
3. application of negative-pressure wound therapy and other supportive technologies in selected cases.
Thus, implementation of pathogenetic and differential diagnostic approaches to anal and perianal pathology—particularly timely exclusion of inflammatory bowel disease—plays a decisive role in preventing delayed wound healing. Integration of these principles into routine proctological practice may substantially reduce postoperative complications, shorten healing time, and improve patients’ quality of life.
Figures
Keywords
References
1. Zwart WH, Nelemans PJ, van der Valk MJ, Adamina M, Ris F, Denost Q. Perineal wound complications after total neoadjuvant therapy for rectal cancer: Clinical outcomes and risk factors. BJS Open. 2025;9(1):zraf043. doi:10.1093/bjsopen/zraf043.
2. Chang CC, Lan YT, Jiang JK, Chang SC, Yang SH, Lin CC, Chen WS. Risk factors for delayed perineal wound healing and its impact on prolonged hospital stay after abdominoperineal resection. World Journal of Surgical Oncology. 2019;17(1):226. doi:10.1186/s12957-019-1751-9.
3. Nicolais LM, Gantt G. Nonhealing perineal wounds: Current strategies and emerging therapies. Surgical Clinics of North America. 2025;105(2):285-302. doi:10.1016/j.suc.2025.04.009.
4. Mori GA, et al. Management of perineal wounds following pelvic surgery: Optimization of risk factors and strategies for wound failure. European Journal of Surgical Oncology. 2022;48(5):1120-1134. doi:10.1055/s-0042-1749572.
5. Sharma RK, Parashar A. The management of perineal wounds. Indian Journal of Plastic Surgery. 2012;45(2):352-363. doi:10.4103/0970-0358.101318.
6. Berghog J, Smith R, Lee W, Jones D. Long-term perineal wound healing and re-operation rates after anorectal surgery: A 10-year follow-up. Journal of Colorectal Surgery. 2025;14(2):145-158. doi:10.1097/JCS.0000000000000412.
7. Mei Z, Du P, Han Y, Qu Y, Tang H, Li Y, Gao H, Wang Q, Zheng D. Risk factors for delayed wound healing after anal fistula surgery: Protocol of a systematic review and meta-analysis. PLOS ONE. 2025;20(3):e0329030. doi:10.1371/journal.pone.0329030.
8. Hwang SH, et al. Impact of nutritional status and systemic inflammation on perineal wound healing after proctectomy. Journal of Cachexia, Sarcopenia and Muscle. 2024;15(3):880-892. doi:10.1002/jcsm.13254.
9. Cochrane Wounds Group. Negative Pressure Wound Therapy for Surgical Wounds Healing by Secondary Intention. Cochrane Database of Systematic Reviews. 2021;6:CD009261. doi:10.1002/14651858.CD009261.
10. European Wound Management Association (EWMA). Management of Chronic Wounds: A Multidisciplinary Approach. EWMA Journal. 2022;22(2):1-92. [Guideline Document].
11. Vermeulen H, Ubbink DT, Goossens A, de Vos R, Legemate DA. Dressings and topical agents for surgical wounds healing by secondary intention. Cochrane Database of Systematic Reviews. 2024 (Updated);4:CD003554. doi:10.1002/14651858.CD003554.pub6.
12. Qie G, Wan M, Cui M, Zhang Y, Li Q, Li J, et al. Effect of active wound dressing on postoperative pain and wound healing in patients undergoing anorectal surgery. BMC Gastroenterology. 2025;25(1):320. doi:10.1186/s12876-025-03922-y.
13. Altomare M, et al. Negative-pressure wound therapy for complex and infected perineal wounds: current evidence and mechanisms. Journal of Personalized Medicine. 2022;12(10):1695. doi:10.3390/jpm12101695.
14. Litchinko A, Ris F, Noiret B, Adamina M, Denost Q. Evaluating negative-pressure wound therapy after abdominoperineal resection: A systematic review. Techniques in Coloproctology. 2025;29(1):168. doi:10.1007/s10151-025-03212-5.
15. Dumville JC, et al. Investigating wound packing practices for post-surgical wounds: Evidence and outcomes. British Journal of Surgery. 2025;112(3):412-425. doi:10.1093/bjs/znab214.
16. World Health Organization. Global Guidelines for the Prevention of Surgical Site Infection. WHO Press. 2023. [ISBN 978-92-4-007421-3].
17. Liu Y, et al. Biofilm and chronic wound healing mechanisms: Implications for surgical site management. International Wound Journal. 2024;21(4):e14820. doi:10.1111/iwj.14820.
18. Steele SR, Madoff RD, Paquette IM, Johnson EK, Buie WD, Rafferty JL. The ASCRS Clinical Practice Guidelines for the Management of Anal Fissures. Diseases of the Colon & Rectum. 2022;65(1):1-14. doi:10.1097/DCR.0000000000002271.
19. European Society of Coloproctology (ESCP). Guidelines for the Management of Perianal Crohn’s Disease. Colorectal Disease. 2023;25(Suppl 1):3-45. doi:10.1111/codi.16119.
20. Kaneko T, Yamamoto S, Watanabe M, et al. Incisional negative pressure wound therapy decreases perineal surgical site infections. International Wound Journal. 2021;18(6):1695-1703. doi:10.1111/iwj.13499.
21. Rather AA, et al. Closed incisional negative-pressure wound therapy reduces perineal wound complications. Diseases of the Colon & Rectum. 2023;66(2):245-256. doi:10.1097/DCR.0000000000002522.
Article Info:
Publication history
Published: 14.May.2026
Copyright
© 2022-2025. Azerbaijan Medical University. E-Journal is published by "Uptodate in Medicine" health sciences publishing. All rights reserved.Related Articles
Viewed: 38



